Capsida Biotherapeutics („Capsida“) today announced the U.S. Food and Drug Administration (FDA) has granted Orphan Drug Designation (ODD) to Capsida for CAP-002. CAP-002 is the company’s lead investigational gene therapy for the treatment of developmental and epileptic encephalopathy (DEE) due to syntaxin-binding protein 1 (STXBP1) mutations. CAP-002 is a novel, first-in-class IV-administered gene therapy using one of Capsida’s proprietary engineered capsids designed to achieve brain-wide neuronal expression of the STXBP1 protein while significantly detargeting the liver. CAP-002 is currently in IND-enabling studies.
FDA Grants Orphan Drug Designation to Capsida Biotherapeutics
LabNews Media LLC
The Editors in Chief of lab-news.de are Marita Vollborn and Vlad Georgescu. They are bestselling authors, science writers and science journalists since 1994.More details about their writing on X-Press Journalistenbüro (https://xpress-journalisten.com).More Info on Wikipedia:About Marita: https://de.wikipedia.org/wiki/Marita_Vollborn About Vlad: https://de.wikipedia.org/wiki/Vlad_Georgescu
Ähnliche Beiträge
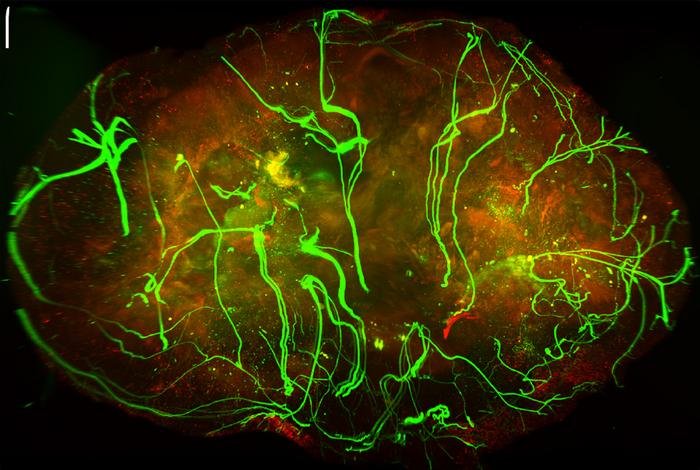
Sympathische Nervenfasern hemmen Wachstum von Melanomen
New York (Labnews Media LLC) – Nervenfasern des sympathischen Nervensystems können das Wachstum von Melanomen verlangsamen. Zu diesem Ergebnis kommt eine Studie der Weill Cornell…
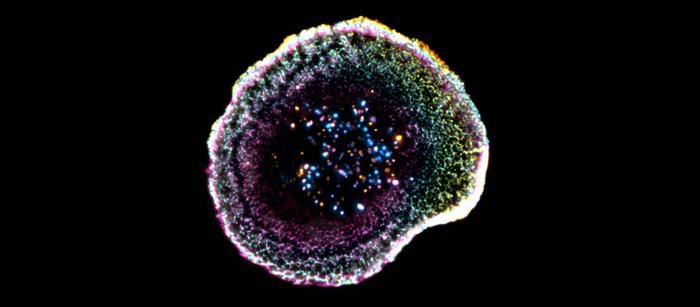
Neue 3D-Technik entschlüsselt die molekulare Organisation der Immunsynapse
Genf/Lausanne (Labnews Media LLC) – Forschende der Universität Genf (UNIGE) und des Universitätsspitals Lausanne (CHUV) haben die feine Struktur der Immunsynapse von zytotoxischen T-Lymphozyten erstmals…

Medizin: Anteil der Diagnosen per IVD
Der Beitrag von IVD (In-vitro-Diagnostika/Labortests) zu Diagnosen und klinischen Entscheidungen ist in fast allen medizinischen Fachgebieten hoch, liegt aber je nach Disziplin zwischen 60 %…
